Sizing up a slow assault on cancer
Rise of immunotherapies spurs search for markers of response.
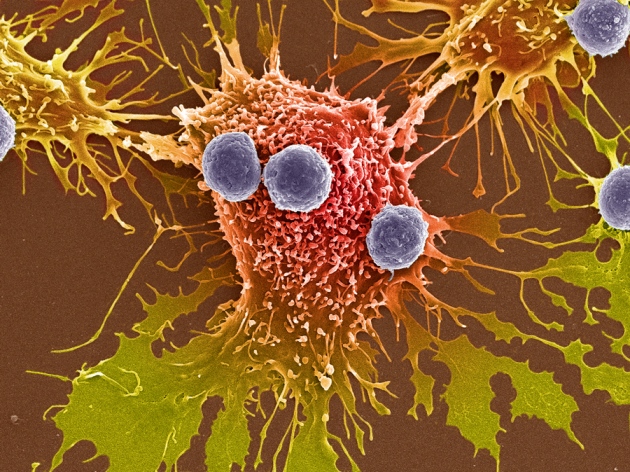
T cells (purple) can signal a slow death for cancer cells (red).
STEVE GSCHMEISSNER/SPL
Jedd Wolchok braced himself as he walked into the examination room to deliver bad news to his patient. Scans showed that the man’s advanced melanoma had spread, and new tumours had sprouted, even though he had received an experimental therapy called ipilimumab (Yervoy) to rally his immune system against the disease. “In my mind I was rehearsing the standard speech,” says Wolchok, an oncologist at the Memorial Sloan-Kettering Cancer Center in New York. “‘I’m sorry Mr So-and-so, but the treatment doesn’t seem to be working.’” Instead, the patient stopped Wolchok at the door. “Now before you show me your pictures, let me tell you: I feel better,” he said.
Wolchok took a gamble, and continued the therapy. More than six years later, his patient is thriving, and the drug, made by Bristol-Myers Squibb of New York, has been approved by the US Food and Drug Administration. In that time, other experimental immunotherapies have shown promise, and clinicians have become increasingly familiar with delayed responses such as the one Wolchok’s patient experienced — which reflect a gradual attack by the immune system, in contrast to the rapid onslaught of toxic chemotherapies. They still have no tools to quickly discern which patients are benefiting from treatment, but that may be about to change.
At the annual meeting of the American Association for Cancer Research in Washington DC this week, Wolchok and other researchers will report on their search for immunotherapy markers — ways to predict a patient’s response to an immunotherapy or to show whether a given treatment is working. The work is hampered by the complexity of the immune system, but early results are converging on one point: that patients’ own immune responses to cancer are crucial in determining outcomes.
“When you sum it all up in many different cancers, early and late stage, it looks like patients who have a ‘smouldering’ initial immune response to cancer are more likely to benefit from immune therapies,” says Thomas Gajewski, a cancer researcher at the University of Chicago in Illinois. Early work suggests that such patients may fare better, irrespective of the therapy used to treat them, he says. The evidence is fuelling a push to add an immune component to standard tumour pathology evaluations (see ‘T cells can brighten prognosis’).
Reliably gauging the initial response, however, is a challenge, cautions Mario Sznol, an oncologist at Yale University in New Haven, Connecticut. The immune response is a moving target, influenced not only by a patient’s genetic make-up and environment, but also by proteins secreted by the tumour that can suppress immune cells. Immune markers present in the blood may not reflect what is happening in the tumour, and variation within a tumour means that biopsies may not paint a full picture either, if the needle misses any patches of immune cells that have infiltrated the tumour.
Some are hoping that new methods will fill the gap. Adaptive Biotechnologies, based in Seattle, Washington, uses intensive DNA sequencing of blood and tissue samples to profile the changes in specialized receptors found in populations of immune cells called T cells. These cells make the receptors by shuffling variable regions of DNA in response to antigen exposure. Chief executive Chad Robins says that the company hopes the assay will be sensitive enough that blood samples alone could be used to monitor patients’ responses to immunotherapy. Demand for the technique has skyrocketed in the past year, he says, as more and more companies seek to test its potential to guide immunotherapy decisions.
“It looks like patients who have a ‘smouldering’ initial response to cancer are more likely to benefit.”
Suzanne Topalian, an oncologist at Johns Hopkins University in Baltimore, Maryland, focuses instead on the cancer cells. A number of the world’s biggest pharmaceutical companies are developing therapies that release a brake on the immune system by inhibiting a protein called programmed death 1 (PD-1) or its partner, programmed death ligand 1 (PD-L1). A study by Topalian and her colleagues1 suggests that these therapies work best if at least 5% of a patient’s biopsied tumour cells express PD-L1. Some companies have already made PD-L1 expression a requirement for participation in their clinical trials — a move that both Topalian and Wolchok worry is premature. “I certainly would not want to exclude a patient from a trial for a PD-1-pathway blocking drug,” says Wolchok. “Even if one tumour biopsy is negative, the patient may have other tumours that are positive.”
While Topalian and others hunt for useful immunotherapy markers, academic labs are trying to understand what ignites the smouldering immune response to cancer in the first place. “Why do some patients have this response when others don’t?” asks Sznol. “Understanding this could lead us to new therapies.”
- Nature
- 496,
- 14–15
- ()
- doi:10.1038/496014a

No comments:
Post a Comment